FOLOTYN Safety Profile
FOLOTYN adverse event profile1,2
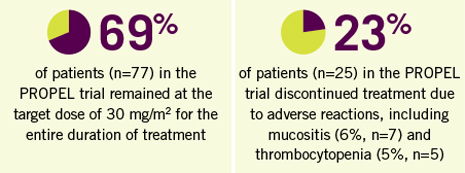
- The most common adverse reactions were mucositis (70%), thrombocytopenia (41%), nausea (40%), and fatigue (36%)
- The most common serious adverse events were pyrexia, mucositis, sepsis, febrile neutropenia, dehydration, dyspnea, and thrombocytopenia
- Incidence of febrile neutropenia: 5% (Grade 3)
- In vitro studies indicated that pralatrexate does not induce or inhibit the activity of CYP450 isozymes at concentrations of pralatrexate that can be reasonably expected clinically
- FOLOTYN is renally excreted—in patients with cancer without renal impairment, approximately 34% of the administered dose of FOLOTYN was excreted unchanged into urine. The mean fraction of the administered dose excreted as unchanged diastereomers in urine (fe) decreased with declining renal function
- When administering FOLOTYN to patients receiving probenecid or other drugs that may affect relevant transporter systems (eg, NSAIDs), monitor patients closely for signs of systemic toxicity due to increased drug exposure
Adverse reactions (incidence ≥10% of patients)1
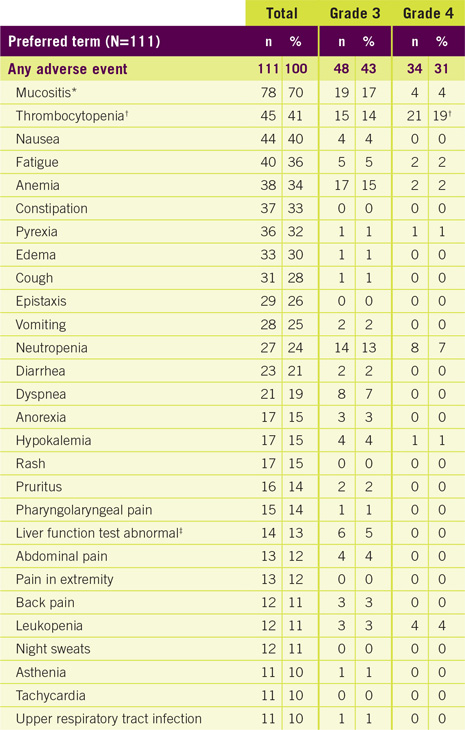
* Stomatitis or mucosal inflammation of the gastrointestinal and genitourinary tracts
† Five patients with platelets <10,000/mcL
‡ Alanine aminotransferase, aspartate aminotransferase, and transaminases increased
References:
-
Folotyn [package insert]. East Windsor, NJ: Acrotech Biopharma, LLC.
-
Data on file. Acrotech Biopharma, LLC.
FOLOTYN Important Safety Information
Indication
FOLOTYN® (pralatrexate injection) is a dihydrofolate reductase inhibitor indicated for the treatment of patients with relapsed or refractory peripheral T-cell lymphoma (PTCL). This indication is approved under accelerated approval based on overall response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
FOLOTYN Important Safety Information
Warnings and Precautions
- Myelosuppression: FOLOTYN can cause myelosuppression, manifested by thrombocytopenia, neutropenia, and/or anemia. Monitor complete blood counts and omit and/or reduce the dose based on ANC and platelet count.
- Mucositis: FOLOTYN can cause mucositis. Monitor for mucositis weekly and omit and/or reduce the dose for ≥ grade 2 mucositis.
- Dermatologic Reactions: Dermatologic reactions, including fatal reactions, occurred and may be progressive and increase in severity with further treatment. Monitor closely and withhold or discontinue FOLOTYN based on severity.
- Tumor Lysis Syndrome: FOLOTYN can cause tumor lysis syndrome. Monitor patients who are at increased risk of TLS and treat promptly.
- Hepatic Toxicity: FOLOTYN can cause hepatic toxicity and liver function test abnormalities. Monitor liver function tests. Omit dose until recovery, adjust or discontinue therapy based on the severity.
- Risk of Increased Toxicity with Renal Impairment: Patients with severe renal function impairment may be at greater risk for increased exposure and adverse reactions. Reduce FOLOTYN dosage in patients with severe renal impairment. Avoid FOLOTYN use in patients with end stage renal disease with or without dialysis. If the potential benefit of administration justifies the potential risk, monitor renal function and reduce the dose based on adverse reactions.
- Embryo-Fetal Toxicity: FOLOTYN can cause fetal harm. Advise patients of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with FOLOTYN and for 6 months after last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with FOLOTN and for three months after last dose.
Adverse Reactions
- The most common adverse reactions occurring in greater than 35% of patients were mucositis (70%), thrombocytopenia (41%), nausea (40%), and fatigue (36%). The most common serious adverse reactions occurring in more than 3% of patients were pyrexia, mucositis, sepsis, febrile neutropenia, dehydration, dyspnea, and thrombocytopenia.
Drug Interactions
- Avoid coadministration with probenecid or nonsteroidal anti-inflammatory drugs (NSAIDs). If coadministration is unavoidable, monitor for increased risk of adverse reactions.
Use in Specific Populations
- Lactation: Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with FOLOTYN and for 1 week after the last dose.
- Pregnancy Testing: Verify pregnancy status in females of reproductive potential prior to initiation of FOLOTYN
- Pediatric Use: The Safety and effectiveness of FOLOTYN in pediatric patients have not been established.
- Renal Impairment: Reduce the dose of FOLOTYN for patients with severe renal impairment (eGFR 15 to 29 mL/min/1.73 m2). No dosage modification is recommended for patients with mild or moderate renal impairment.
Please click here to see full Prescribing Information for FOLOTYN.
ISI-FOL-0180


